An injection-based treatment tested in mice turns immune cells into a weapon against cancer
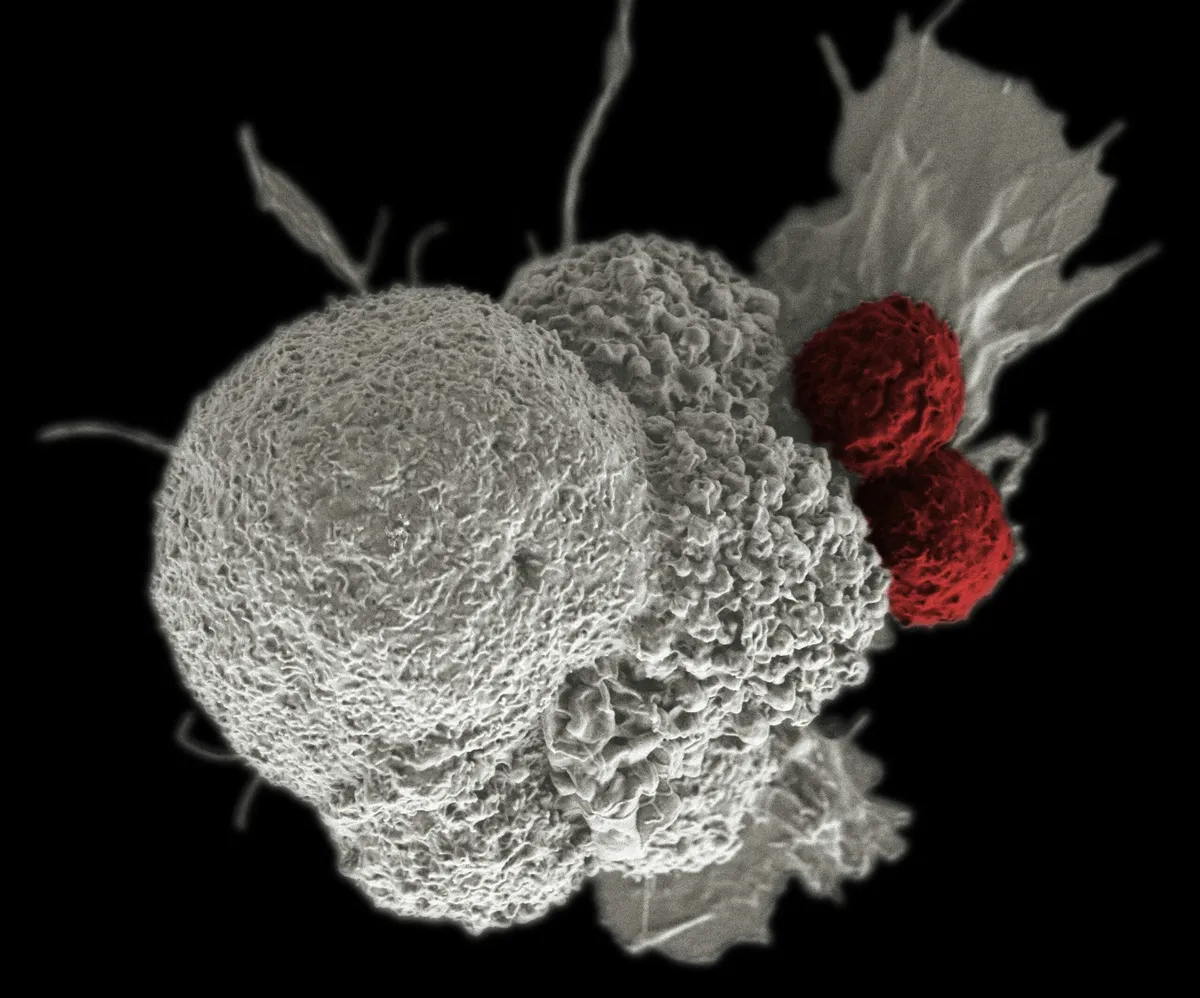
Researchers at the University of California, San Francisco (UCSF), led by Associate Professor Justin Eyquem, have developed a gene‑therapy technique that can reprogramme immune cells directly inside the body. This could make treatments cheaper and more widely available, particularly for blood cancers.
CAR‑T therapies have for years changed care for some blood cancers by modifying a patient’s own immune cells so they recognise and kill tumour cells. The current approach, however, involves a complex, slow and costly manufacturing chain that requires specialised facilities and keeps treatment confined to large cancer centres. Eyquem and his team say their technique can make CAR‑T cells inside the body, which could cut costs and shorten waiting times.
Where CAR‑T therapy stands now
Traditional CAR‑T therapy follows a few precise steps. Doctors first remove T lymphocytes from the patient, white blood cells involved in immune defence. Those cells are taken to a lab and genetically altered to include a Chimeric Antigen Receptor (CAR), which lets them spot and attack cancer cells. Before the modified cells go back into the patient, chemotherapy is used to prepare the bone marrow.
The whole process takes weeks and is very expensive: between £312,000 and £390,000 per patient in the United States. The need for specialised facilities also limits who can access the treatment. There are safety concerns too: the random insertion of the CAR gene during modification could lead to secondary cancers. Although there are seven FDA‑approved CAR‑T therapies, they all target blood cancers and so far have not worked against solid tumours.
Making CAR‑T cells inside the body (in vivo)
The method from UCSF uses a two‑particle system to generate CAR‑T cells in vivo (inside the body). One particle delivers the CRISPR‑Cas9 gene‑editing system, which targets T lymphocytes circulating in the blood. The other carries DNA that encodes the CAR receptor. By inserting the CAR DNA at a precise spot in the genome, this approach avoids the risks tied to random gene insertion seen with older techniques. Eyquem says the cells edited in the body “seem even better than those we manufacture in the laboratory.”
Preclinical trials in mice support these findings. A single injection wiped out detectable leukaemia in most of the mice tested within a fortnight. The approach also worked against multiple myeloma and showed promise against solid tumours such as sarcoma, areas where standard treatments struggle. The CRISPR‑guided precision and the added safety from targeted insertion may offer advantages over current options.
What this could mean for cancer care
If these results translate to humans, the in vivo method could reduce the financial and logistical burden of CAR‑T therapies and might allow community hospitals to offer them rather than relying on major cancer centres. To move toward human trials, Eyquem’s team has launched Azalea Therapeutics. Full commercial roll‑out could still take several years because of the clinical testing required, but the work at UCSF indicates a different approach to CAR‑T delivery.
The study was published in Nature on 18 March, and the collaboration includes teams from UCSF, the Gladstone Institutes, Duke University, and the Innovative Genomics Institute. Jennifer Doudna, a Nobel laureate and co‑founder of the Innovative Genomics Institute, plays a leading role in advancing this research.
The medical and social effects of such a development could include wider access to effective cancer treatments across more patient groups.